I’m super early. Day surgery unit is in darkness! BUT admiring the metal work and fabulous stained glass windows behind – feel I need to stay here until it’s light to see the windows better!
Tag Archives: Phyllodes
Surgery Prep
Surgery was scheduled for next week for boob op. I passed all the tests (well apart from high white blood count) and even my ecg. BUT today they managed to get ecgs from my “a&e adventure” last week!
They’ve tried all day to speak to the cardiologist at the other hospital to pursuade him to see me quickly but blocked from even speaking to him!
<<Meanwhile (I didn’t know about this glitch) so I cancelled a small op planned on my ankle. That’ll be rescheduled for February next year… or later! >>
Sooo at about 5 tonight they made the decision to cancel my op until the cardiologist has seen me/more tests etc. So pissed off. Just want it all to be over.
Taking bets on surgery being scheduled for my 50th birthday!
Post scan results
Seen consultant for results of last weeks scan. Operation in the next few months to remove stupid lump…
I’ve asked him to put a zip in so it’s easier if any others pop up later! Hey ho.
Biopsy Results
My biopsy results have been reviewed by Cyril (superstar path at Royal Marsden) and discussed at the MDT meeting along, apparently, with lengthy discussion about phyllodes and our online support group!!!
Decision remains that the biopsy showed benign fibroadenoma. HOWEVER because of my history and weirdness of phyllodes they’ve decided to bring me back in September to rescan, measure and possibly biopsy (although if it’s grown, they may just do surgery and not bother with biopsy again). So all good. Thanks to those asking. I’m quite chilled about it all. I have the bestest consultant and team at the Marsden and I know if I was worrying they’d bring me in sooner. So for now just lots of bubbles required…. always fizz!!
Scan time
Results
Results time for my 6 monthly scans. If there’s nothing to report, they say I may go to annual checks!!!
UPDATE
No Evidence of Disease again… Finally I‘m on annual checks! AND my fave consultant made sure he saw me to give me the good news… AND now he’s head of the sarcoma unit here, he’s writing something for our Phyllodes Support Group AND he suggested I get my MRI and X-ray for my odd ankle to him to review. Hurrah!! Calls for bubbles and opera!
Scan time
Time for my 6 monthly mammogram ultrasound and chest X-ray. Already had mammogram. F’ing hurt and that was after 4 Nurofen! All results in a few weeks!
Oh and in other other news the fracture clinic at another hospital have lost my X-rays!
Investigations
For over a year I’ve not felt brilliant with what I’ll describe (to save your blushes) with an upset stomach. I’ve felt nauseous and at times been sick. I won’t bore you with lots of other symptoms but suffice to say I did bore my GP with them. Several visits and told I must have IBS or perhaps eaten something that didn’t agree. Eventually I was referred to a gastro specialist but the referral day was when there was a computer glitch so required following up, running around picking up letters and chasing phone calls. I eventually saw a specialist on 9th September. Between the last visit to the GP and my consultant appointment I had noticed a small lump below my rib cage but typically I couldn’t feel it when I was in the appointment. He urgently referred me for a colonoscopy and ultrasound.
I got to wear one of those lovely gowns and a pair of paper pants with a velcro flap at the back! Hahaha so gorgeous. Oh and how ironic that as I walked back through the waiting room, guess who was on the tellybox?
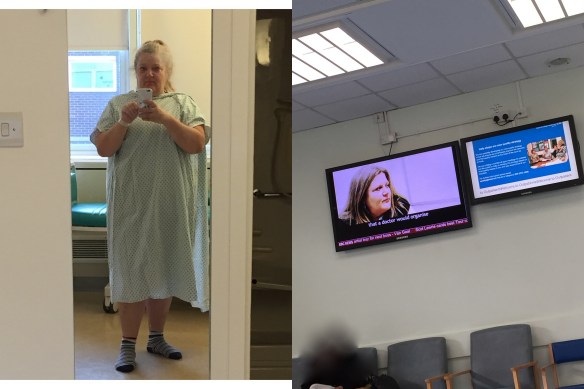 They took a number of biopsies during the procedure but couldn’t find the lump and suggested it’s in the connective tissue. Maybe they’ll work it out from the ultrasound. Although that then doesn’t explain my upset stomach and nausea.
They took a number of biopsies during the procedure but couldn’t find the lump and suggested it’s in the connective tissue. Maybe they’ll work it out from the ultrasound. Although that then doesn’t explain my upset stomach and nausea.
6 monthly scan results
A couple of weeks ago I visited the Royal Marsden for my Summer 6 month scans – chest x-ray, ultrasound and boob squish. Stupidly (and I should know better) I neglected to take some ibuprofen before my boob squish. I was once again in extreme pain and so super envious of ladies who don’t have pain. (How do some ladies go jogging without a secure bra?) I suspected the mammographer had spotted something as she insisted on doing a couple of extra scans to ‘get in the sides’.
Next up was my ultrasound. Whilst I was changing into my ‘everso sexy’ dressing gown, I heard the radiographer being called in to see my mammogram results. Long story short she said she could see a small dot but suspected it is a cyst.
Today was my appointment with my consultant. He has vast experience with phyllodes tumours as well as a wonderful disposition and honest, caring approach to me, the patient. I’m so very grateful for such a fabulous consultant.
We talked about ‘the dot’. We’re not sure it’s a cyst. I’ve had no other cysts before now. BUT rather than poke around and upset my breast tissue, we decided to leave it ‘just so’. My consultant has asked that in six months we have a full set of scans and tests again (usually I have a mammogram once a year in the Summer). He did, however say that if I had any concerns or worries at all, I was to contact him directly and an appointment would be found. I’m happy with this. I appreciate his experience and the open discussion. I feel assured I’m in the best hands and care.
He also made sure we spoke about my auto-immune disease. The drugs I had taken and the side affects I’m still left with (as well as the residual disease).
We spoke about my friend who’s currently undergoing surgeries following the discovery of new phyllodes tumours in her heart, liver and lung. He wants me to forward details of the hospital and surgeon so he can learn more about the case and educate himself and his team.
We also spoke about a clinical trial that I’m trying to get set up in the UK for Phyllodes. I’ll write more about it soon… hopefully with good news. Once again, he wants me to keep him in the picture so that he can ensure he/the Royal Marsden are able to recruit for the trial. Such positive news, we hope.
Finally we spoke about life. His and mine. He asked me how I was doing. Was there anything else I was worried about. Any other aches, pains or points of concern.
I feel totally looked after… and hopefully the dot will remain just that, a dot.
Glass Memories of Washington
When I visited Washington at Easter, we (Andi, Sam and I) chose an object that I could use to create something in glass. The object we chose was a pine cone. You’d think that’d be simple to use. Perhaps to create the object in glass? Perhaps use the texture from the cone? Or something else? When I showed the tutor she dismissed creating it in glass as the cone was open and difficult to cast. She suggested I use the base to create a latex mold and then add these ‘flowers’ to a piece.
Over several weeks, I’ve been making the wax ‘flowers’. I decided that I’d like to do a bowl with the flowers around it.
The process is the same, create the was ‘flowers’, create a wax bowl, affix the ‘flowers’ securely and try to close up the gaps to reduce risk with plaster. It takes an age to get the completed wax model.
Then I mixed up the plaster and flint mix to pour over the wax model. Next stage is to steam out the wax. I purchased some emerald green gaffer glass which flows faster than bullseye glass. I then handed it over to the technicians to fire in the kiln.
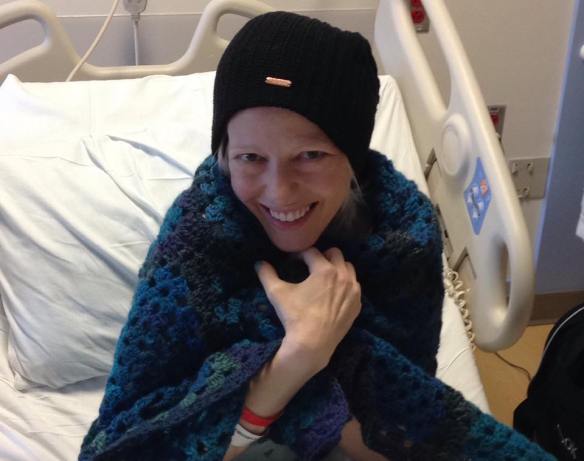
All the above I did without telling Andi how I was creating the piece. I was excited to create it and then present it to Andi as a completed piece in the future. In June I’d not heard from Andi for a few days and was worried. A few days later she messaged me from the hospital… and told me she was wrapped in her AnnaBlanket which made me smile.
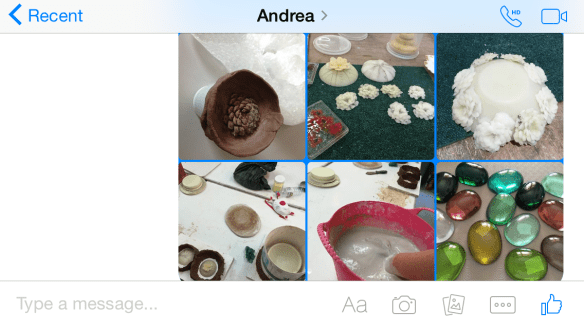
 And I let her into the secret and told her what I was doing with the pine cone and a few photos.
And I let her into the secret and told her what I was doing with the pine cone and a few photos.
 After all that work and with the terrifically important reason I was making this piece, the pressure was on for this to be perfect. Only a few days later I was at college and the piece was cooling in the kiln. I could tell that something had happened as the technicians asked me to look in the kiln. The mold had cracked and glass leaked. So incredibly upset.
After all that work and with the terrifically important reason I was making this piece, the pressure was on for this to be perfect. Only a few days later I was at college and the piece was cooling in the kiln. I could tell that something had happened as the technicians asked me to look in the kiln. The mold had cracked and glass leaked. So incredibly upset.
 I love that damaged imperfect bowl. It’s not useable and needs a lot of work to clean up or reuse the glass. Right now, I can’t do anything with it except be disappointed and sad. Maybe one day soon I’ll create something with the glass that will work and be an Andi treasure.
I love that damaged imperfect bowl. It’s not useable and needs a lot of work to clean up or reuse the glass. Right now, I can’t do anything with it except be disappointed and sad. Maybe one day soon I’ll create something with the glass that will work and be an Andi treasure.



